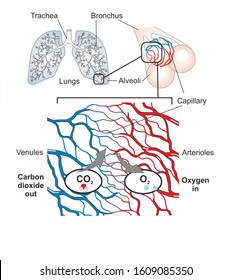
Recall that the respiratory system works to humidify incoming air, thereby causing the air present in the alveoli to have a greater amount of water vapor than atmospheric air. The amount of water vapor present in alveolar air is greater than that in atmospheric air (Table 2). In both cases, the relative concentration of gases is nitrogen > oxygen > water vapor > carbon dioxide. The composition of air in the atmosphere and in the alveoli differs. Gas molecules establish an equilibrium between those molecules dissolved in liquid and those in air. Too much nitrogen in the bloodstream results in a serious condition that can be fatal if not corrected. The exception to this occurs in scuba divers the composition of the compressed air that divers breathe causes nitrogen to have a higher partial pressure than normal, causing it to dissolve in the blood in greater amounts than normal. For example, although nitrogen is present in the atmosphere, very little nitrogen dissolves into the blood, because the solubility of nitrogen in blood is very low. The concentration of the gas in a liquid is also dependent on the solubility of the gas in the liquid. The greater the partial pressure of the gas, the greater the number of gas molecules that will dissolve in the liquid. Henry’s law states that the concentration of gas in a liquid is directly proportional to the solubility and partial pressure of that gas. Henry’s law describes the behavior of gases when they come into contact with a liquid, such as blood. In addition, the greater the partial pressure difference between the two areas, the more rapid is the movement of gases. A gas will move from an area where its partial pressure is higher to an area where its partial pressure is lower. 
Recall that gases tend to equalize their pressure in two regions that are connected. Partial pressure is extremely important in predicting the movement of gases. The sum of the partial pressures of all the gases in a mixture equals the total pressure. Partial pressure is the force exerted by a gas. Dalton’s law describes the behavior of nonreactive gases in a gaseous mixture and states that a specific gas type in a mixture exerts its own pressure thus, the total pressure exerted by a mixture of gases is the sum of the partial pressures of the gases in the mixture. 
Total pressure is the sum of all the partial pressures of a gaseous mixture. For example, in the atmosphere, oxygen exerts a partial pressure, and nitrogen exerts another partial pressure, independent of the partial pressure of oxygen (Figure 1). Partial pressure ( P x) is the pressure of a single type of gas in a mixture of gases. Total composition/total atmospheric pressure For example, the atmosphere consists of oxygen, nitrogen, carbon dioxide, and other gaseous molecules, and this gaseous mixture exerts a certain pressure referred to as atmospheric pressure (Table 1). In natural systems, gases are normally present as a mixture of different types of molecules. Gas molecules exert force on the surfaces with which they are in contact this force is called pressure.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
